What is Healthcare Environmental Monitoring?
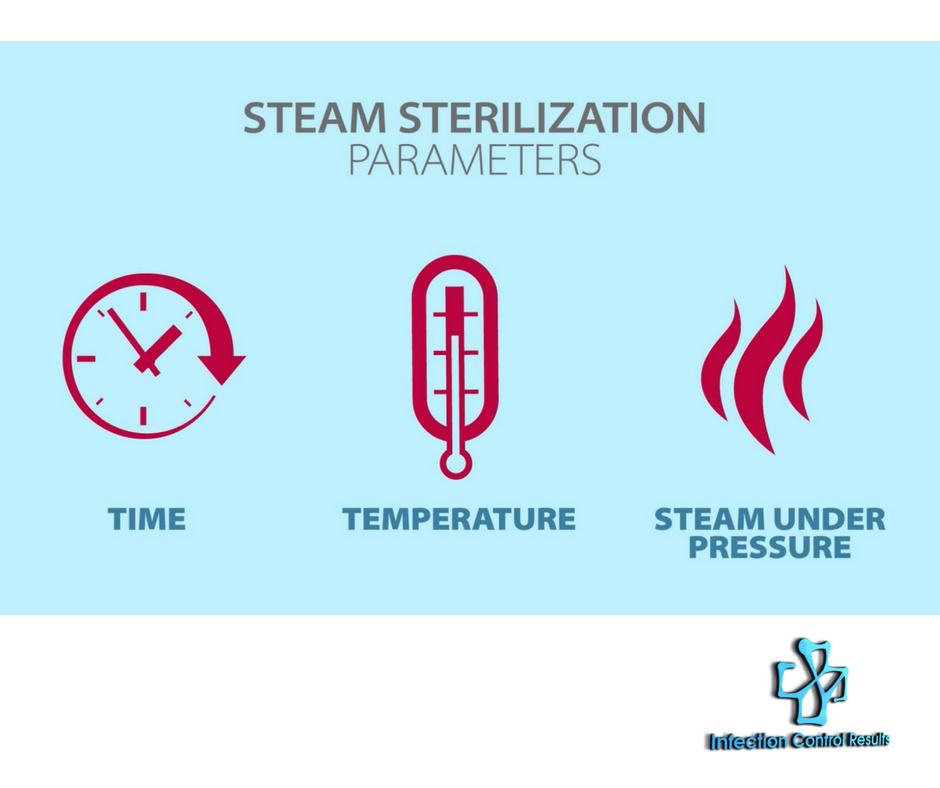
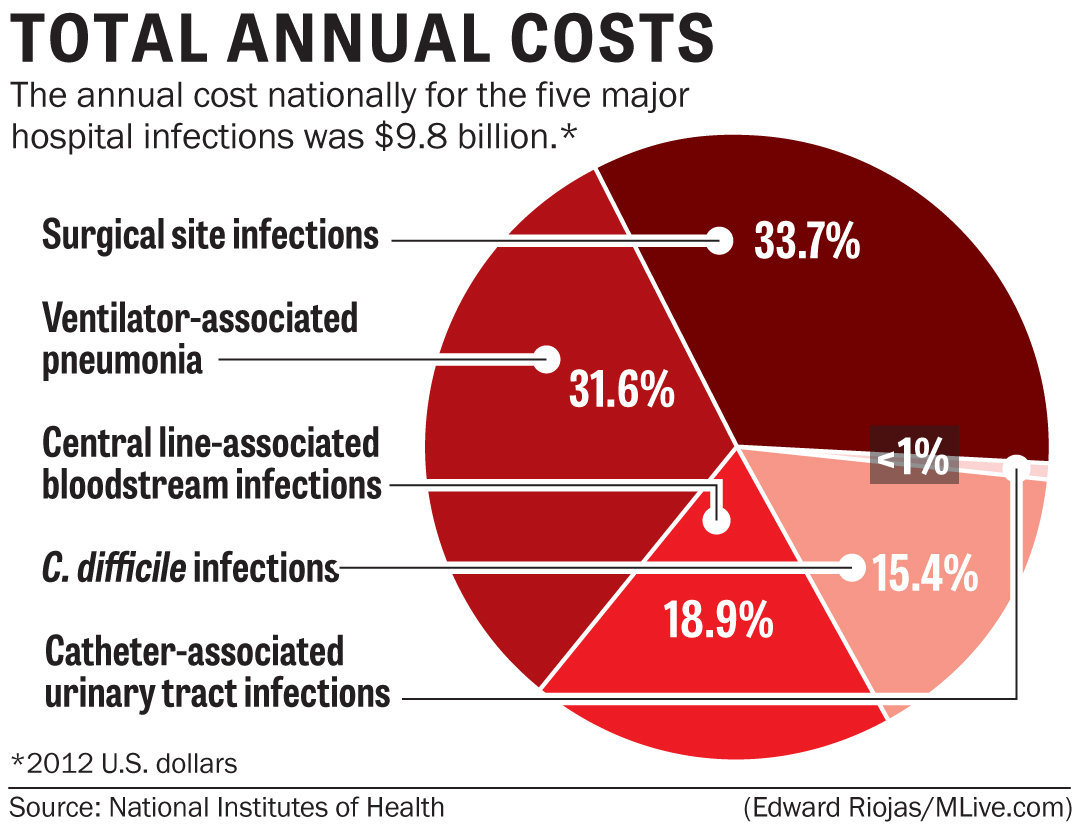
When we think of environmental monitoring, temperature, humidity, and air exchanges in the operating room (OR) and sterile processing department (SPD) usually come to mind. However, there are numerous other areas in both acute and ambulatory care settings that must be monitored. Environmental Monitoring Basics Accrediting and...